Categories
Tags
-
#ENT Devices Market
#Precision Medicine Market
#ENT Devices Market report
#ENT Devices industry
#Dental Radiography Market
#Dental Radiography Market report
#Dental Radiography industry
#NGS Sample Preparation Market
#NGS Sample Preparation Market report
#NGS Sample Preparation industry
#Precision Medicine Market report
#Precision Medicine industry
#Operating Room Integration Systems Market
#Operating Room Integration Systems Market report
#Operating Room Integration Systems industry
#Middle East Precision Medicine Market
#Middle East Precision Medicine Market report
#Middle East Precision Medicine industry
#Digital Biomarkers Market
#Digital Biomarkers Market report
#Digital Biomarkers industry
#Lung Cancer Genomic Testing Market
#Lung Cancer Genomic Testing Market report
#Lung Cancer Genomic Testing industry
#Precision Medicine Market size
#Precision Medicine Market research
#Precision Medicine Market value
#Precision Medicine Market share
#Precision Medicine Market drivers
#Capillary Electrophoresis Market
#Capillary Electrophoresis Market report
#Capillary Electrophoresis industry
#Capillary Electrophoresis size
#Gene Cloning Services Market
#Gene Cloning Services Market report
#Gene Cloning Services industry
#Gene Cloning Services Market size
#Gene Cloning Services Market value
#Gene Cloning Services Market research
#brain disease modalities and software market report
#brain disease modalities and software market
#brain disease modalities and software industry
#Middle East Liquid Biopsy Market
#Middle East Liquid Biopsy Market Report
#Middle East Liquid Biopsy Industry
#Middle East Liquid Biopsy Market size
#Middle East Liquid Biopsy Market value
#Aseptic Pharma Processing Market
#Aseptic Pharma Processing Market Report
#Aseptic Pharma Processing Industry
#Aseptic Processing Market
#Non-Invasive Prenatal Testing (NIPT) Market
#Non-Invasive Prenatal Testing (NIPT) Market report
#Non-Invasive Prenatal Testing (NIPT) Industry
#Non-Invasive Prenatal Testing (NIPT) Market size
#Viral and Non-Viral Vector Manufacturing Market
#Non-Viral Vector Manufacturing Market
#Viral and Non-Viral Vector Manufacturing Market Report
#Viral and Non-Viral Vector Manufacturing Industry
#Viral Vector Manufacturing Market
#Carrier Screening Market
#Carrier Screening Market Report
#Carrier Screening Industry
#Carrier Screening Market size
#Epigenetics Market
#Epigenetics Market Report
#Epigenetics Industry
#Epigenetics Market size
#Epigenetics Market Value
#Epigenetics Market Research
#Cancer Microbiome Sequencing Market
#Cancer Microbiome Sequencing Market Report
#Cancer Microbiome Sequencing industry
#Cancer Microbiome Sequencing Market size
#Cancer Microbiome Sequencing Market share
#Molecular Diagnostics Market
#Molecular Diagnostics Market Report
#Molecular Diagnostics industry
#Molecular Diagnostics Market size
#Molecular Diagnostics Market share
#Molecular Diagnostics Market trends
#Europe Hereditary Genetic Testing Market
#Europe Hereditary Genetic Testing Market Report
#Europe Hereditary Genetic Testing industry
#Europe Genetic Testing Market
#Europe Hereditary Genetic Testing Market size
#Organ Transplant Diagnostics Market
#Organ Transplant Diagnostics Market report
#Organ Transplant Diagnostics industry
#Organ Transplant Diagnostics Market size
#Organ Transplant Diagnostics Market value
#Organ Transplant Market
#Biomaterials Market
#Biomaterials Market report
#Biomaterials industry
#Biomaterials Market size
#Biomaterials Market value
#Biomaterials Market share
#Nucleic Acid Therapeutics CDMO Market
#Nucleic Acid Therapeutics CDMO Market report
#Nucleic Acid Therapeutics CDMO industry
#Liquid Biopsy Market
#Liquid Biopsy Market report
#Liquid Biopsy industry
#Liquid Biopsy Market size
#Liquid Biopsy Market value
#Liquid Biopsy Market research
#Liquid Biopsy Market share
#Liquid Biopsy Market analysis
#Liquid Biopsy Market growth
#Middle East Molecular Diagnostics Market
#Middle East Molecular Diagnostics Market report
#Middle East Molecular Diagnostics industry
#Middle East Molecular Diagnostics Market value
#Middle East Molecular Diagnostics Market growth
#Molecular Diagnostics Market research
#Single Cell RNA Sequencing Market
#Single Cell RNA Sequencing Market report
#Single Cell RNA Sequencing industry
#Single-cell Analysis Market
#Single Cell Sequencing Market
#OR Management Solutions Market
#OR Management Solutions Market report
#OR Management Solutions industry
#OR Management Solutions Market trends
#OR Management Solutions Market size
#OR Management Solutions industry value
#brain imaging modalities market report
#brain imaging modalities market
#brain imaging modalities industry
#brain imaging modalities market size
#Spine X-Ray and Computed Tomography (CT) Market
#Spine X-Ray and Computed Tomography (CT) Market report
#Spine X-Ray and Computed Tomography (CT) industry
#Next-Generation Gynecological Cancer Diagnostics Market
#Next-Generation Gynecological Cancer Diagnostics Market report
#Next-Generation Gynecological Cancer Diagnostics industry
#Next-Generation Gynecological Cancer Diagnostics Market Size
#MRD Testing Market
#MRD Testing Market report
#MRD Testing industry
#MRD Testing Market size
#MRD Testing Market value
#MRD Testing Market growth
#U.S. Solid Tumor Testing Market
#U.S. Solid Tumor Testing Market report
#U.S. Solid Tumor Testing industry
#U.S. Solid Tumor Testing Market size
#U.S. Solid Tumor Testing Market value
#U.S. Solid Tumor Testing Market growth
#U.S. Solid Tumor Testing Market research
#Minimally Invasive Surgical Systems Market
#Minimally Invasive Surgical Systems Market report
#Minimally Invasive Surgical Systems Industry
#Minimally Invasive Surgical Systems Market size
#Lung Cancer Genomic Testing Market size
#Lung Cancer Genomic Testing Market trends
#Precision Cardiology Market
#Precision Cardiology Market report
#Precision Cardiology industry
#Precision Cardiology Market size
#Precision Cardiology Market value
#Precision Cardiology Market growth
#Big Data in Healthcare Market
#Big Data in Healthcare Market report
#Big Data in Healthcare industry
#Big Data in Healthcare Market size
#Big Data in Healthcare Market value
#Big Data in Healthcare share
#Axial Spondyloarthritis Market
#Axial Spondyloarthritis Market report
#Axial Spondyloarthritis industry
#Axial Spondyloarthritis Market size
#axial spondyloarthritis treatment market
#Medical Grade Tubing Market
#Medical Grade Tubing Market report
#Medical Grade Tubing Industry
#Medical Grade Tubing Market size
#Medical Grade Tubing Market value
#Medical Grade Tubing Market growth
#Preimplantation Genetic Testing Market
#Preimplantation Genetic Testing Market report
#Preimplantation Genetic Testing industry
#Preimplantation Genetic Testing Market size
#Preimplantation Genetic Testing Market value
#Preimplantation Genetic Testing Market growth
#Pharmaceutical Quality Management Software Market
#Pharmaceutical Quality Management Software Market Report
#Pharmaceutical Quality Management Software industry
#Pharmaceutical Quality Management Software Market size
#Pharmaceutical Quality Management Software Market value
#mRNA Vaccines and Therapeutics Market
#mRNA Vaccines and Therapeutics Market report
#mRNA Vaccines and Therapeutics industry
#mRNA Vaccines and Therapeutics Market size
#Europe Infectious Disease Diagnostics Market
#Europe Infectious Disease Diagnostics Market report
#Europe Infectious Disease Diagnostics industry
#Europe Infectious Disease Diagnostics Market size
#Europe Infectious Disease Diagnostics Market growth
#NGS Oncology Market
#NGS Oncology Market report
#NGS Oncology Market industry
#Clinical Oncology Next Generation Sequencing Market
#Blood and Plasma Components Market
#Blood and Plasma Components Market Report
#Blood and Plasma Components industry
#Blood Components Market
#Blood and Plasma Components Market size
#Blood and Plasma Components Market share
#Pharmacogenomics Services Market
#Pharmacogenomics Services industry
#Pharmacogenomics Services Market report
Archives
Europe Infectious Disease Diagnostics Market Analysis
-
Posted by BIS Reports - Filed in Business - #Europe Infectious Disease Diagnostics Market #Europe Infectious Disease Diagnostics Market report #Europe Infectious Disease Diagnostics industry #Europe Infectious Disease Diagnostics Market size #Europe Infectious Disease Diagnostics Market growth - 1,004 views
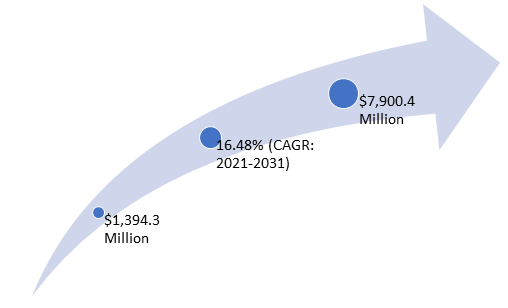
The use of infectious disease diagnostics kits, assays, and other consumables has increased due to the rising prevalence of cancer, the increasing key player initiatives, and the rising government initiatives. The increasing investments by healthcare companies to meet industry demand and the growing adoption of infectious disease diagnostics kits, tubes, and reagents among major end users are major factors propelling the growth of the Europe infectious disease diagnostics market. The Europe infectious disease diagnostics market was valued at $1,394.3 million in 2020 and is expected to reach $7,900.4 million by 2031, witnessing a CAGR of 16.48% during the forecast period 2021-2031.
Competitive Landscape
The Europe infectious disease diagnostics market comprises well-established and newly emerging companies. Several companies are attempting to sustain their position in the market by launching new products and raising funds to develop new products for infectious disease diagnostics from different product types. Key market players of the Europe infectious disease diagnostics market witnessed product launch and approval, business funding, and synergistic activities from January 2018 to March 2022. The inclination of companies toward product launches suggests that companies are constantly bringing new products to the market to test viral and bacterial infections, which is primarily attributed to an increase in demand for kits and assays by end users.
Market Segmentation
• Product (system, kits and reagents, and software)
• Testing location (laboratory testing and point-of-care testing)
• Technology (next-generation sequencing (NGS), polymerase chain reaction (PCR), immunodiagnostics, isothermal nucleic acid amplification technology (INAAT), microarray, in-situ hybridization (ISH), and other technologies)
• Infectious Disease Type (bacterial infectious diseases, viral infectious diseases, fungal infectious diseases, and other infectious diseases)
• Infection Type (respiratory infections, hospital-acquired infections (HAI), sexually transmitted infections (STIs), and other infections)
• End User (hospitals, diagnostic centers, out-patient clinics/general practitioners, research laboratories, and other end users)
Regional Segmentation
• Europe-5 - Germany, U.K., France, Italy and Spain
• Nordic- Denmark, Sweden, Norway and Finland
• Baltic- Estonia, Lithuania and Latvia
• Rest-of-EuropeEurope Infectious Disease Diagnostics Market Dynamics

Key Questions Answered in this Report:
• How is each segment of the market expected to grow during the forecast period 2021-2031, and what is the anticipated revenue to be generated by each segment? Following are the segments:
o Product (system, kits and reagents, and software)
o Testing location (laboratory testing and point-of-care testing)
o Technology (next-generation sequencing (NGS), polymerase chain reaction (PCR), immunodiagnostics, isothermal nucleic acid amplification technology (INAAT), microarray, in-situ hybridization (ISH), and other technologies)
o Infectious Disease Type (bacterial infectious diseases, viral infectious diseases, fungal infectious diseases, and other infectious diseases)
o Infection Type (respiratory infections, hospital-acquired infections (HAI), sexually transmitted infections (STIs), and other infections)
o End User (hospitals, diagnostic centers, out-patient clinics/general practitioners, research laboratories, and other end users)
o Region (EU-5, Baltic, Nordic, and Rest-of-Europe)
• What are the major market drivers, challenges, and opportunities in the Europe infectious disease diagnostics market?
• What are the underlying structures resulting in the emerging trends within the Europe infectious disease diagnostics market?
• How is each segment of the Europe infectious disease diagnostics market expected to grow during the forecast period, and what will be the expected revenue generated by each of the segments by the end of 2031?
• What are the key developmental strategies implemented by the major players to sustain in the competitive market?
• What are the key regulatory implications in developed and developing regions for molecular diagnostics?
• Who are the leading players with significant offerings to the Europe infectious disease diagnostics market? What is the current market dominance for each of these leading players?
• What would be the compound growth rate witnessed by the leading players in the market during the forecast period 2021-2031? Which infectious disease product type is anticipated to have the most promising growth?
• What are the key applications in the Europe infectious disease diagnostics market? What are the major segments of these applications? What technologies are dominating these application segments?
• What are the major technologies that are employed in the Europe infectious disease diagnostics market? Which is the dominating technology?
• Who are the primary end users of the Europe infectious disease diagnostics market? Which is the fastest-growing end-user segment in the Europe infectious disease diagnostics market?
• Who are the key manufacturers in the Europe infectious disease diagnostics market, and what are their contributions? Also, what is the growth potential of each major infectious disease diagnostics manufacturer?
• What is the scope of the Europe infectious disease diagnostics market in the EU’5, Baltic, and Nordic regions? Which infectious disease diagnostics product and end user dominate these regions?
• What are the emerging trends in the Europe infectious disease diagnostics market? How are these trends revolutionizing the diagnostic procedure?
• Which technologies are anticipated to break through the current infectious diseases diagnostic regime?
• Which companies are anticipated to be highly disruptive in the future and why?
• What are the regulatory procedures required to unify the approval process for emerging infectious disease diagnostics? How will these enhance the reimbursement scenario?
• What are the gaps in regularizing optimum infectious disease diagnostics adoption in regular healthcare routines? How are these gaps being tackled?Request Sample - https://bisresearch.com/requestsample?id=1294type=download
BIS Related Studies

